Elemental Detection of Semiconductor Process Gases Using a Gas-Exchange Device Coupled to ICP-MS
This article describes the direct measurement of trace levels of organometallic process gases in semiconductor manufacturing environments using a gas-exchange device coupled with quadrupole inductively coupled plasma–mass spectrometry (ICP-MS).
This article describes the direct measurement of trace levels of organometallic process gases in semiconductor manufacturing environments using a gas-exchange device coupled with quadrupole inductively coupled plasma–mass spectrometry (ICP-MS). By passing ambient air through the gas-exchange device, the organometallic gas molecules in the sample are quantitatively extracted and transferred to the ICP-MS system for detection as the metallic species. The combined system and analysis approach are summarized, and fully quantitative calibrations are included.
Organometallic gases are used in a variety of manufacturing processes in the semiconductor industry. For example, gases such arsine (AsH3) and germane (GeH4) are used in metalorganic chemical vapor-phase deposition (MOCVD) processes to produce thin layers in complex semiconductor devices.
The use of these gases in manufacturing processes is complicated by their toxicity. For example, exposure to arsine may lead to renal failure and it is a recognized carcinogen. Although arsine can be detected by its garlic-like odor at concentrations above 0.5 ppm, that level is well above the current American Conference of Governmental Industrial Hygienists (ACGIH) threshold limit value (TLV) of 0.005 ppm over an 8-h period.
Although alternatives to the use of organometallic gases in semiconductor manufacturing processes are being assessed, significant steps must be taken to avoid accidental exposure of personnel to these gases. Accepted methods for the routine monitoring of arsine in the workplace environment involve trapping it on a sorbent material, digestion, and subsequent analysis by graphite furnace atomic absorption spectrometry (GF-AAS) or inductively coupled plasma–optical emission spectrometry (ICP-OES). These methods, however, cannot provide the real-time results necessary for a meaningful assessment of current workspace conditions.
This article describes an alternative technique for the direct measurement of trace amounts of semiconductor gases in ambient air using a quadrupole ICP–mass spectrometry (MS) system coupled with a gas-exchange device (GED). Although ICP-MS is recognized as an extremely sensitive elemental detector, it is commonly used for the analysis of solutions or of solid samples after they have been brought into solution. The GED overcomes problems in the direct elemental analysis of air by ICP-based techniques by exchanging atmospheric gases with argon, which is compatible with the ICP ion source. Direct air sampling using the GED and on-line elemental analysis by ICP-MS therefore provides immediate information on airborne contamination.
Principle of the Gas-Exchange Device
The GED (J-Science Lab Co. Ltd.) consists of a concentric glass tube inside a glass (Pyrex, Corning) body (Figure 1). Pores, 0.07 µm in diameter, in the inner glass tube allow it to act as a membrane between the gaseous sample passing through the inner central channel of the GED and argon in the surrounding outer channel.
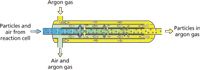
Figure 1: Schematic of the gas-exchange device (GED).
The combination of the GED with an ICP-MS system has been previously demonstrated for the multielemental monitoring of particles in environmental atmospheric gases (1,2). In the semiconductor process gas application described here, the target analytes (Ge and As) are found in their gaseous states (GeH4 and AsH3, respectively) in the workplace environment. Before entry into the GED, gaseous analytes in the air sample must be converted into particles so that they are not lost across the porous membrane. A reaction cell is used to generate suitable particles from the sampled gas before entry into the GED. Ozone in the reaction cell converts metal hydrides to their respective oxides via the following reactions:
GeH4 (g) + 2O3 → GeO2 + 2H2O + O2
2AsH3 (g) + 2O3 → As2O3 + 3H2O
A 2% ammonia solution (prepared using Optima-grade ammonia, Thermo Scientific) is used to generate ammonia gas that reacts with the ozone inside the reaction cell to generate nitrate particles via the following reaction:
2NH3 + 4O3→ NH4NO3 + H2O + 4O2
The AsO3 and GeO2 nanoparticles agglomerate with NH4NO3 in the reaction cell to form particles too large to pass through the porous membrane of the GED. As the gas with suspended particles travels along the inner tube of the GED, atmospheric gases are replaced with argon via diffusion across the membrane. Ultimately, before the sample enters the plasma, the atmospheric gases have been replaced by argon and the particulate matter resulting from the gas or air sample remains. The NH4NO3 particles are easily dissociated in the argon plasma into their constituent ions while the As2O3 and GeO2 provide a stable signal suitable for quantification by ICP-MS.
Experimental
Instrument Configuration
Routine analysis with the reaction cell–GED–ICP-MS system consists of manually introducing individual air samples (standards and so forth) directly into the Sample Gas In port of the reaction cell–GED device. Samples are collected by syringe and are stored in 1-L Tedlar (DuPont) gas sampling bags before introduction into the reaction cell–GED. Individual analyses (blanks, samples, and standards) are therefore processed manually with each introduction of air corresponding to an individual ICP-MS acquisition.
A schematic of the GED is shown in Figure 1. The particle-supporting argon gas stream from the reaction cell is connected directly to the ICP-MS system's (iCAP Qs, Thermo Scientific) sapphire injector using a ball-joint connector. Nitrogen make-up gas (supplied by the reaction cell–GED) was added immediately before the sapphire injector using a glass T-piece to further improve sensitivity. Platinum-tipped sampler and skimmer cones were used.
The ICP-MS system was operated in a single analysis mode for the measurement of both 74Ge and 75As. A 3 V kinetic energy discrimination (KED) barrier with pure He as collision gas in the iCAP Q QCell collision–reaction cell was used to suppress the formation of any interfering polyatomic species at the target masses. The operating parameters used for the reaction cell-GED and ICP-MS systems used in this work are shown in Table I.
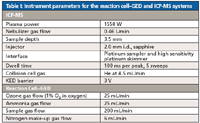
Table I: Instrument parameters for the reaction cellâGED and ICP-MS systems
Standards at concentrations of 5, 10, 25, 500, 1000, and 2500 pptv (parts per trillion by volume) for GeH4 and 1, 2, 5, 100, 200, and 500 pptv for AsH3 were prepared by dilution with 99.9995% pure nitrogen in clean Tedlar bags from a mixed GeH4–AsH3 gas standard (Gastec Corporation).
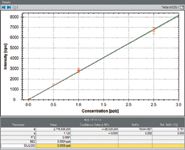
Figure 2: Calibration curve for GeH4 from 5 to 2500 pptv.
The ICP-MS system was optimized for maximum sensitivity using a gas mixture of Cr(CO)6, Mo(CO)6, and W(CO)6 generated by an element standards gas generator (ESGG, J-Science Lab Co. Ltd.). The standards gas generator produces a continuous stream of elemental vapor in an argon gas flow that is introduced directly into the ICP-MS system, bypassing the reaction cell–GED system.
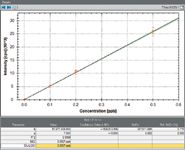
Figure 3: Calibration curve for AsH3 from 1 to 500 pptv.
Results and Discussion
Linear calibrations for GeH4 and AsH3 (measured at masses 74Ge and 75As, respectively) were obtained and are displayed in Figures 2, 3, and 4. Each point on the calibration curve is generated from three separate measurements. Figure 3 shows the calibration for As across the entire concentration range, and Figure 4 shows the same calibration for concentrations lower than 10 pptv to demonstrate the high sensitivity of the technique, with 1 pptv of As recording a sensitivity of 50 cps.
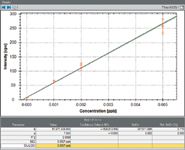
Figure 4: Calibration curve for AsH3 from 1 to 5 pptv.
Instrumental limits of detection limits (LOD) were calculated as three times the standard deviation from 10 replicate measurements of 99.9995% nitrogen. A background equivalent concentration (BEC) was calculated for Ge and As from the nitrogen. The LOD and BEC values obtained are shown in Table II.
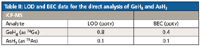
Table II: LOD and BEC data for the direct analysis of GeH4 and AsH3
A simple recovery test using a 500 pptv GeH4 and 100 pptv AsH3 standard was performed, and recoveries of 98% for GeH4 and 105% for AsH3 were obtained. Precisions (% relative standard deviation [RSD]) of 3.3% for GeH4 and 2.6% for AsH3 were obtained in this analysis.
Nitrogen is commonly used in dry plasma applications (for example, laser ablation and desolvating nebulizer–based analyses) to improve ionization in the argon ICP ion source. The resulting sensitivity improvement is element specific and dependent on the first ionization of the element (or elements) being measured. For the determination of As with a relatively high first ionization potential of 9.8 eV, the use of nitrogen can lead to significant sensitivity improvements. The use of nitrogen to improve sensitivity with the reaction cell–GED–ICP-MS system was assessed, and the results are shown in Table III.
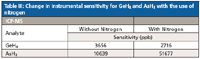
Table III: Change in instrumental sensitivity for GeH4 and AsH3 with the use of nitrogen
Whereas a fivefold improvement in sensitivity was seen for AsH3 when using additional nitrogen, there was no appreciable improvement for GeH4.
To assess any memory effect from the reaction cell–GED sampling system, a pure nitrogen gas blank was introduced directly after the highest concentration standard and the Ge and As intensities were monitored. Within 6 min the 74Ge and 75As count rates returned to the gas blank levels, indicating the absence of any significant memory effect. At the trace concentrations found in real samples, analysis times (including uptake and washout) are 2 min. The conversion in the reaction cell takes less than 30 s.
Because the analysis is equivalent to manual sample introduction in solution mode by ICP-MS, the full range of quality control features (calibration and blank verification for example) can be implemented as part of the analysis sequence. In the same way, samples above defined exposure limits can be automatically flagged for rapid action as necessary.
Although current methodology requires manual point sample introduction, automated gas sampling and introduction into the reaction cell–GED–ICP-MS system are under development for improved sample throughput. In situ long-term sampling is possible but wasn't tested as part of this initial study.
Conclusion
A highly sensitive and specific method for the direct analysis of trace amounts of GeH4 and AsH3 semiconductor process gases in ambient air has been demonstrated. With this new approach, the presence of these highly toxic gases can be quickly and accurately determined, leading to improved worker safety in manufacturing environments.
References
(1) Y. Suzuki, H. Sato, K. Hiyoshi, and N. Furuta, Spectrochimica Acta Part B 76, 133–139 (2012).
(2) T. Vincent, K. Nishiguchi, K. Utani, S. McSheehy Ducos, and L. Rottmann, AN43170 (Thermo Scientific, 2013).
Tomoko Vincent, Julian D. Wills, and Lothar Rottmann are with Thermo Fisher Scientific in Bremen, Germany. Kohei Nishiguchi and Keisuke Utani are with J-Science Lab Co. in Kyoto, Japan. Direct correspondence to: julian.wills@thermofisher.com

Inside the Laboratory – The Petrochronology Group at University of California, Santa Barbara
January 29th 2024In this edition of “Inside the Laboratory,” John Cottle, PhD, a professor of geology at the University of California, Santa Barbara, and a member of Spectroscopy’s Editorial Advisory Board, discusses his group’s most recent work using “laser ablation split steam” analysis to measure elemental concentrations and isotopic ratios in rocks and minerals.