Single Reaction Chamber Microwave Digestion for Specialty Chemicals
Application Notebook
Single reaction chamber microwave digestion enables a chemist to digest up to 15 different specialty chemical samples simultaneously at temperatures as high as 300 ?C, greatly simplifying the workflow while maintaining superior quality digestions.
Single reaction chamber microwave digestion enables a chemist to digest up to 15 different specialty chemical samples simultaneously at temperatures as high as 300 °C, greatly simplifying the workflow while maintaining superior quality digestions.
The class of "Specialty Chemicals" encompasses a wide variety of chemicals ranging from single chemical entities to complex formulations. While some of these chemicals are easy to prepare for trace metals analysis, many others require extremely high temperatures for complete digestion, which is difficult to achieve even with conventional closed vessel microwave systems. Often chemists have to complement microwave technology with traditional tools like hot plates and Paar bombs to digest the highly stable matrices which in turn have their own set of limitations — large acid requirements, contamination, acid handling challenges, lengthy digestions cycles, and exposure of chemists to acid fumes. Finally, although multiple samples can be digested in closed vessel microwaves simultaneously, samples of similar matrices need to be batched and run separately to ensure complete control over the system. This can severely hamper the productivity of a lab testing specialty chemicals, as a wide variety of sample matrices often require preparation.
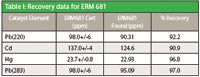
Table I: Recovery data for ERM 681
Milestone's UltraWAVE SRC benchtop digestion system can digest up to 15 different samples simultaneously at temperatures and pressures as high as 300 °C and 199 bar. This high temperature and pressure capability enables a complete digestion of almost any specialty chemical that needs to be analyzed for trace metals. Samples can be directly weighed into disposable glass, autosampler type vials with the appropriate acid mixtures; no minimum acid quantity is required. Quartz and TFM vials can also be used depending on the user's application. This minimizes acid handling and transferring steps, which reduces errors due to contamination and the chemist's exposure to acids. It also completely eliminates the need to clean, assemble, and disassemble vessels used in conventional microwave systems.
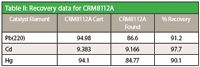
Table II: Recovery data for CRM8112A
Experiment
The reaction chamber was pre-pressurized to 40 bar to prevent the acids from boiling, which subsequently prevented cross-contamination and loss of volatiles. 15-position racks were used with disposable glass vials which were covered by loose fitting caps. This ensures pressure equilibrium on either side of the vial while preventing the condensation from the top of the chamber from dripping into the sample. 0.2g of ERM681 (low density polyethylene) and CRM8112A (ABS Resin) were simultaneously digested in 4 ml of 68% nitric acid. The samples were heated to 225 °C in 15 min and held at 225 °C for 15 min. The trace metals in the digested samples were analyzed using a PE Optima ICP-OES system.
The data shows excellent DLs and recoveries. No loss of volatile elements (like Hg) or cross contamination was observed. Due to higher sample capacity, use of disposable vials and faster cool down time, the UltraWAVE sample throughput is 2–3 times higher as compared to closed vessel microwave digestions. Its lower consumables costs, simple operation, and superior digestion quality make Milestone's UltraWAVE a perfect choice for specialty chemical's sample prep.
Milestone, Inc.
Controls Drive, Shelton, CT 06484
tel. (866) 995-5100, fax (203) 925-4241
Website: www.milestonesci.com

Single Cell and Microplastic Analysis by ICP-MS with Automated Micro-Flow Sample Introduction
April 25th 2024Single cell ICP-MS (scICP-MS) is increasingly seen as a powerful and fast tool for the measurement of elements in individual cells, mainly due to the high sensitivity and selectivity of ICP-MS. Analysis is performed in the same way as single nanoparticle (spICP-MS) analysis, which has become a well-established technique for the analysis of nanoparticles and particles.
Hot News on Agilent LDIR, New Developments, and Future Perspective
April 25th 2024Watch this video featuring Darren Robey and Dr. Wesam Alwan from Agilent Technologies to gain insights into the future trends shaping microplastics research and the challenges of their characterization. Discover the essential components necessary for accurate microplastics analysis and learn how the Agilent 8700 LDIR system addresses these challenges. Offering rapid and precise analysis capabilities, along with easy sample preparation methods that minimize contamination, the Agilent 8700 LDIR system is at the forefront of advancing microplastics research.
The World of Microplastics Up to Date – an Overview
April 23rd 2024Watch this 20-minute educational video by Andreas Kerstan, Agilent Product Specialist in molecular spectroscopy, to gain a comprehensive update on the microplastics landscape and the environmental concerns related to them. Discover the current challenges in microplastics characterization and how Agilent innovative solutions and techniques, including FTIR, LDIR, GC/MS, and ICP-MS, are addressing these issues head-on.