Analyzing Highly Fluorescing Cellulose-Based Excipients and Other Complex Molecules with a Novel 1030 nm Handheld Raman Analyzer
Handheld Raman analyzers have found widespread use in the pharmaceutical industry for excipients and raw materials verification, and in first responder/security applications. Most portable Raman units utilize lasers operating at 785 nm because of the relatively good signal strength at this wavelength.
Handheld Raman analyzers have found widespread use in the pharmaceutical industry for excipients and raw materials verification, and in first responder/security applications. Most portable Raman units utilize lasers operating at 785 nm because of the relatively good signal strength at this wavelength.
There are many compounds that fluorescence strongly at 785 nm, causing a featureless background spectrum that obscures the telltale Raman signature. Compounds include the micro-crystalline cellulose (MCC) compounds used as excipients, and nutrients like Folic acid (vitamin B9). For security applications, Semtex and TNT precursors are good examples.

Figure 1, 2: Raman spectra for material MCC Generic (top) and Croscarmellose (bottom) comparing a 785 nm laser and a 1030 nm laser. The Raman peak structure in both compounds is only evident when utilizing the 1030 nm laser.
SciAps has introduced a handheld Raman analyzer utilizing a novel 1030 nm laser source – SWIR Raman. The 1030 nm laser yields reduced fluorescence compared to 785 nm lasers. The 1030 nm laser is compatible with a proprietary Type III-IV sensor that requires less cooling than analyzers using 1064 nm laser sources. There are no cooling fans required, and smaller batteries may be used. Therefore a 1030 nm system is very portable, and is sealed to IP67 standards, making it dunkable and washable for quarantine or hotzone use.
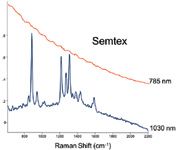
Figure 3: Raman spectrum from a common plastic explosive Semtex. The characteristic Raman peak structure is only evident when using the 1030 nm laser compared to the 785 nm.
One Example: MCC Generic and Croscarmellose
MCC is a particularly good example. MCC can be modified to different degrees of crystalinity usually in the range of 40 to 60%. Raman spectra from MCC generic and Croscarmellose, using the 785 nm laser (Inspector 300) and the 1030 nm laser (Inspector 500) are shown in Figures 1 and 2. The Raman peaks of the MCC and Croscarmellose clearly stand out at 1030 nm but are lost in the featureless fluorescence background at 785 nm. A similar benefit is shown for plastic explosive Semtex. Semtex is a combination of PETN and cyclonite (cyclotrimethylenetrinitramine) that contains complex double bonded N, C, and O structures that produce fluorescence at 785 nm laser wavelengths. As shown in Figure 3 this material is easily identified with a 1030 nm laser source.
SciAps, Inc.
2 Constitution Way, Woburn, MA 01801
tel. (339) 927-9455
Website: www.sciaps.com

The Effect of Heat Exposure on BPA and Phthalate Content in Commercial Bottled Water
May 8th 2024This study aimed to assess the levels of phthalates and bisphenol A (BPA) in various popular bottled waters compared to tap water. Additionally, it investigated whether exposure to high temperatures, akin to those in a car during summer, would elevate these levels. Using GC/MS analysis, samples were examined. Results showed that phthalates and BPA were either absent or well below safety thresholds in all bottled and tap water samples. Moreover, heating did not significantly affect phthalate concentrations, and BPA was undetectable in all samples.
Spex Speaks Science: Soil and Contamination
May 8th 2024The science and art of gardening and agriculture is part passion, part luck and a lot of chemistry. In this podcast, we will take a look at the science of soil and how chemistry can affect the growth and health of plants. We will also take a look at sources of potential contamination in gardens and how those contaminants can be tested and remediated. Hosted by Patricia Atkins, Manager, Global Product at Spex®.
The Analysis of Laboratory and Consumer Water Sources for the Presence of BPA and Phthalates
May 8th 2024Bottled water has evolved from a trendy luxury to a global consumer staple, with significant growth projected by 2012. In the U.S., consumption has soared, with safety concerns and substitution driving its popularity. Developing nations also rely on bottled water for safety. This study aims to address debated issues surrounding BPA and phthalate exposure in consumer water sources: 1. Are BPA and phthalates present in bottled water? 2. Does exposure to high temperatures increase leaching of BPA or phthalates? 3. Are levels in municipal or filtered water significantly different from bottled water?