Temperature Induced Changes in Clays Measured by Diamond ATR FT-IR Spectroscopy
Application Notebook
Increasing temperature can cause chemical changes, alter reaction rates, concentrate an aqueous sample, or evaporate a solvent. These transformations can be readily monitored by diamond ATR spectroscopy. This note explores the use of a diamond ATR to examine heat-induced changes in the clay.
Increasing temperature can cause chemical changes, alter reaction rates, concentrate an aqueous sample, or evaporate a solvent. These transformations can be readily monitored by diamond ATR spectroscopy. This note explores the use of a diamond ATR to examine heat-induced changes in the clay.
Experimental
The measurements were carried out using a diamond single reflection ATR, the MVP-Pro™ with its heated diamond crystal holder and temperature controller (Figure 1) installed in a commercial FT-IR spectrometer equipped with a DTGS detector. The temperature controller was autotuned for operation to 200 °C. Then the desired set point was selected as needed. Spectra were collected at an 8 cm-1 resolution, a gain of 8, and signal averaged over 32 scans. A small mound of the montmorillonite ("otay"; SCa-3, Clay Minerals Society) was pressed against the diamond ATR using the maximum force delivered by the built-in pressure applicator.

Figure 1: MVP-Pro Star shown with heated cell and temperature controller.
Results and Discussion
Figure 2 shows the spectra of otay before and after heating. Clays such as otay are hygroscopic and easily absorb water at room temperature. The resulting water peak in the 3000 cm-1 to 3400 cm-1 region can obscure the characteristic C–H stretch. Heating the sample to 200 °C for 5 min drives off the water, making the C–H peaks in these samples emerge or become more distinct. In all of the figures, blue signifies room temperature spectra while red denotes heated spectra. The portion of the spectrum in the 2500 cm-1 to 2000 cm-1 region is not shown due to the absorbance of diamond in that region.
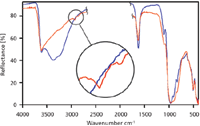
Figure 2: Otay spectra at 26 °C (blue) and 200 °C (red).
Conclusion
Heated diamond ATR accessories can be used effectively to examine evaporation upon heating. Diamond ATR accessories such as the MVP-Pro™ are powerful tools for examining heat-induced chemical transformations.
Harrik Scientific Products, Inc.
141 Tompkins Ave., P.O. Box 277, Pleasantville, NY 10570
tel. (800) 248-3847
Website: www.harricksci.com

The Effect of Heat Exposure on BPA and Phthalate Content in Commercial Bottled Water
May 8th 2024This study aimed to assess the levels of phthalates and bisphenol A (BPA) in various popular bottled waters compared to tap water. Additionally, it investigated whether exposure to high temperatures, akin to those in a car during summer, would elevate these levels. Using GC/MS analysis, samples were examined. Results showed that phthalates and BPA were either absent or well below safety thresholds in all bottled and tap water samples. Moreover, heating did not significantly affect phthalate concentrations, and BPA was undetectable in all samples.
Spex Speaks Science: Soil and Contamination
May 8th 2024The science and art of gardening and agriculture is part passion, part luck and a lot of chemistry. In this podcast, we will take a look at the science of soil and how chemistry can affect the growth and health of plants. We will also take a look at sources of potential contamination in gardens and how those contaminants can be tested and remediated. Hosted by Patricia Atkins, Manager, Global Product at Spex®.
The Analysis of Laboratory and Consumer Water Sources for the Presence of BPA and Phthalates
May 8th 2024Bottled water has evolved from a trendy luxury to a global consumer staple, with significant growth projected by 2012. In the U.S., consumption has soared, with safety concerns and substitution driving its popularity. Developing nations also rely on bottled water for safety. This study aims to address debated issues surrounding BPA and phthalate exposure in consumer water sources: 1. Are BPA and phthalates present in bottled water? 2. Does exposure to high temperatures increase leaching of BPA or phthalates? 3. Are levels in municipal or filtered water significantly different from bottled water?