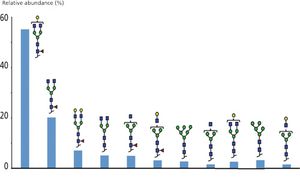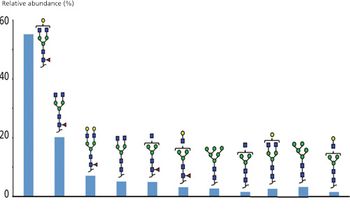
Antibody drug conjugates (ADCs) are an emerging category of biotherapeutic products based on monoclonal antibodies (mAbs) coupled to powerful cytotoxic drugs. The production of ADCs entails the formation of species with different number of conjugates drugs. The heterogeneity of ADCs species add to the complexity originating from the mAbs microvariability. Sheathless capillary electrophoresis-mass spectrometry (sheathless CE-MS) using complementary approaches was used to perform a detail characterization of brentuximab vedotin (Adcetris, Seattle Genetics). Sheathless CE-MS instrument used as nanoESI infusion platform was involved to perform the intact and middle-up analysis in native MS conditions. The nanoESI infusion approaches enabled estimation of the average drug to antibody ratio (DAR) alongside to drug load distribution. Sheathless CZE-MS/MS method developed was used to obtain from a single injection the characterization of the amino acid sequence with complete sequence coverage. In addition glycosylation and drug-loaded peptides could be identified from MS/MS spectra revealing robust information regarding their localizations and abundances. Drug-loaded peptide fragmentation mass spectra study demonstrated drug-specific fragments reinforcing the identifications confidence. Results reveal the ability of sheathless CZE-MS/MS method to characterize ADCs primary structure in a single experiment.