Spectroscopy
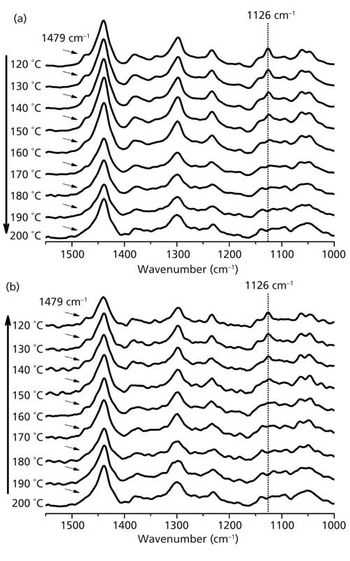
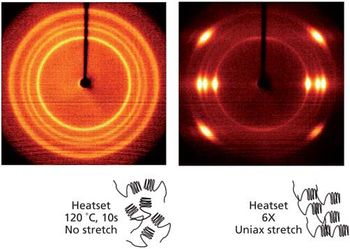
In the “Molecular Spectroscopy Workshop” column, we have been trying to provide hands-on advice and easy-to-implement tips to start using spectroscopic tools for the analyst who has the responsibility to derive answers to questions as quickly as possible. Very often the identity of an unknown is of ultimate importance, and very few analytical chemists coming out of graduate programs have been taught to systematically analyze spectra to infer identity of the source. In addition, it is rare that an industrial environment will provide resources for the analyst to be educated in this field. Thus, the availability of any means to provide spectral identification will make the difference between success and failure; that is, acquiring a spectrum is useless if one cannot identify it. In this column, I discuss some of the fundamentals of spectral interpretation, illustrate the use of searching software, including mixture analysis, and show how sometimes the software can provide spectral interpretation.