Keith C. Gordon
Articles by Keith C. Gordon

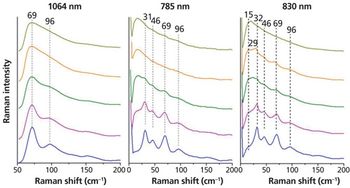
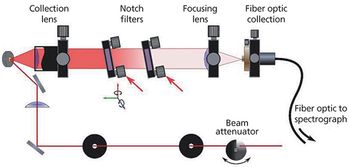
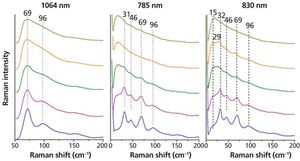
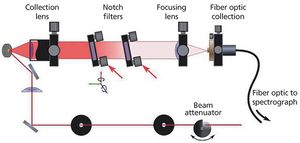
Crystallinity is an important factor when producing pharmaceuticals as it directly affects the bioavailability of the drug. Low frequency Raman spectroscopy offers some advantages to the detection and analysis of crystallinity in pharmaceutical samples. Here the experimental requirements for low frequency Raman measurements are described. The application to the study of crystallinity with a number of examples is discussed and the advantages and limitations of this technique are highlighted and compared with other techniques.

The authors show how a multivariate curve resolution algorithm, called SIMPLe-to-use Interactive Self-modeling Mixture Analysis (SIMPLISMA), can facilitate the quantitative and qualitative analysis of difficult samples, and apply the algorithm to a technically challenging Raman spectra series for carbamazepine polymorphs.
Latest Updated Articles
 How Using Raman Spectroscopy and SIMPLISMA Can Accelerate the Study of Polymorphs: A Case Study Using Carbamazepine (PDF)
How Using Raman Spectroscopy and SIMPLISMA Can Accelerate the Study of Polymorphs: A Case Study Using Carbamazepine (PDF)Published: June 2nd 2004 | Updated:
 Investigating Crystallinity Using Low Frequency Raman Spectroscopy: Applications in Pharmaceutical Analysis
Investigating Crystallinity Using Low Frequency Raman Spectroscopy: Applications in Pharmaceutical AnalysisPublished: February 1st 2016 | Updated:
 Investigating Crystallinity Using Low-Frequency Raman Spectroscopy: Applications in Pharmaceutical Analysis
Investigating Crystallinity Using Low-Frequency Raman Spectroscopy: Applications in Pharmaceutical AnalysisPublished: February 1st 2017 | Updated: