
In this article, we introduce the concept of a gas exchange device (GED) and how it can be used to monitor organometallic compounds and metallic particles in specialty gases.

In this article, we introduce the concept of a gas exchange device (GED) and how it can be used to monitor organometallic compounds and metallic particles in specialty gases.
Special Issues
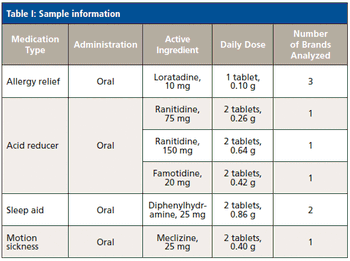
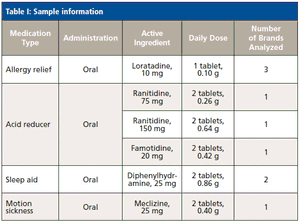
With its advantages-compared to ICP-MS-in matrix tolerance, robustness, simplicity, and cost, ICP-OES may be the technique of choice for the analysis of medications with low daily doses. For such analyses, the use of high-pressure, high-temperature microwave digestion in sealed containers simplifies sample preparation and prevents the loss of volatile elements.
Spectroscopy
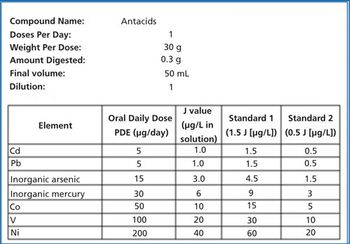
Antacids present a unique set of analytical challenges for ICP-MS. These challenges can be overcome with optimized sample preparation and instrumental analytical conditions.
September 1st 2018